Membres
Projets
Agnès LEHUEN
Jacques BELTRAND
Le rôle des lymphocytes T de type inné dans la régulation du Diabète de Type 1
Michel POLAK
Aurore CARRE
Développement et fonction de la thyroïde
Publications
Les dernières actualités
Brevets et réseaux
BREVETS
European patent “Methods and kits of assessing status, risk or prognosis of type 1 diabetes”
EP16 305 319.2 on March 22 2016
PCT/EP2017/056859 on March 22 2017 (WO2017/162759) via le site espacenet de l’OEB.
US2019011432 (A1) on 2019-01-10
Lehuen, J. Da Silva, I. Nel, L. Beaudoin
European patent « Methods and pharmaceutical compositions for the treatment of fibrosis with agents capable of inhibiting the activation of mucosal-associated invariant T (MAIT) cells »
EP2017/051669 on June 22 2017 (WO2018/234843) via le site espacenet de l’OEB.
US2021403573 (A1) on 2021-12-30
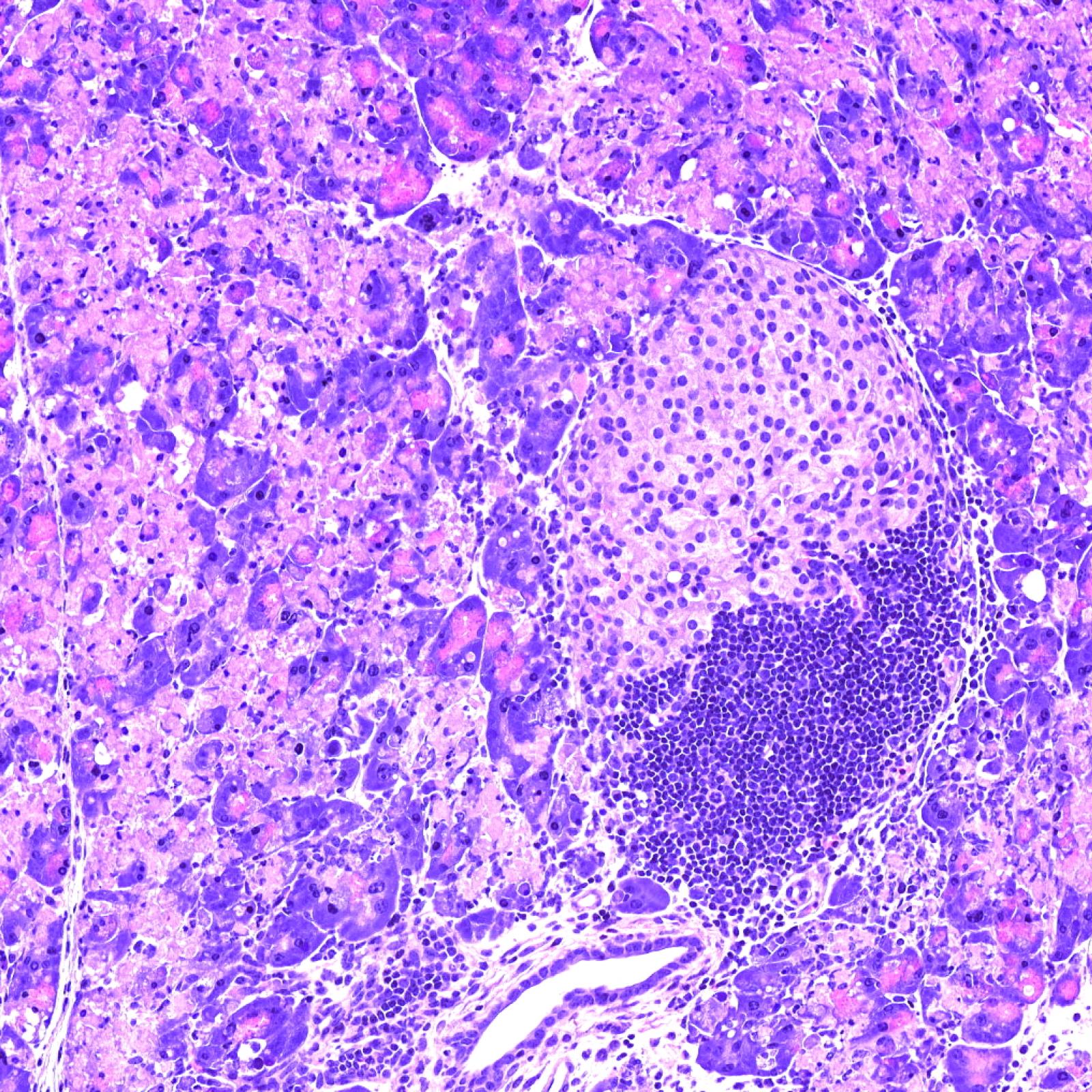
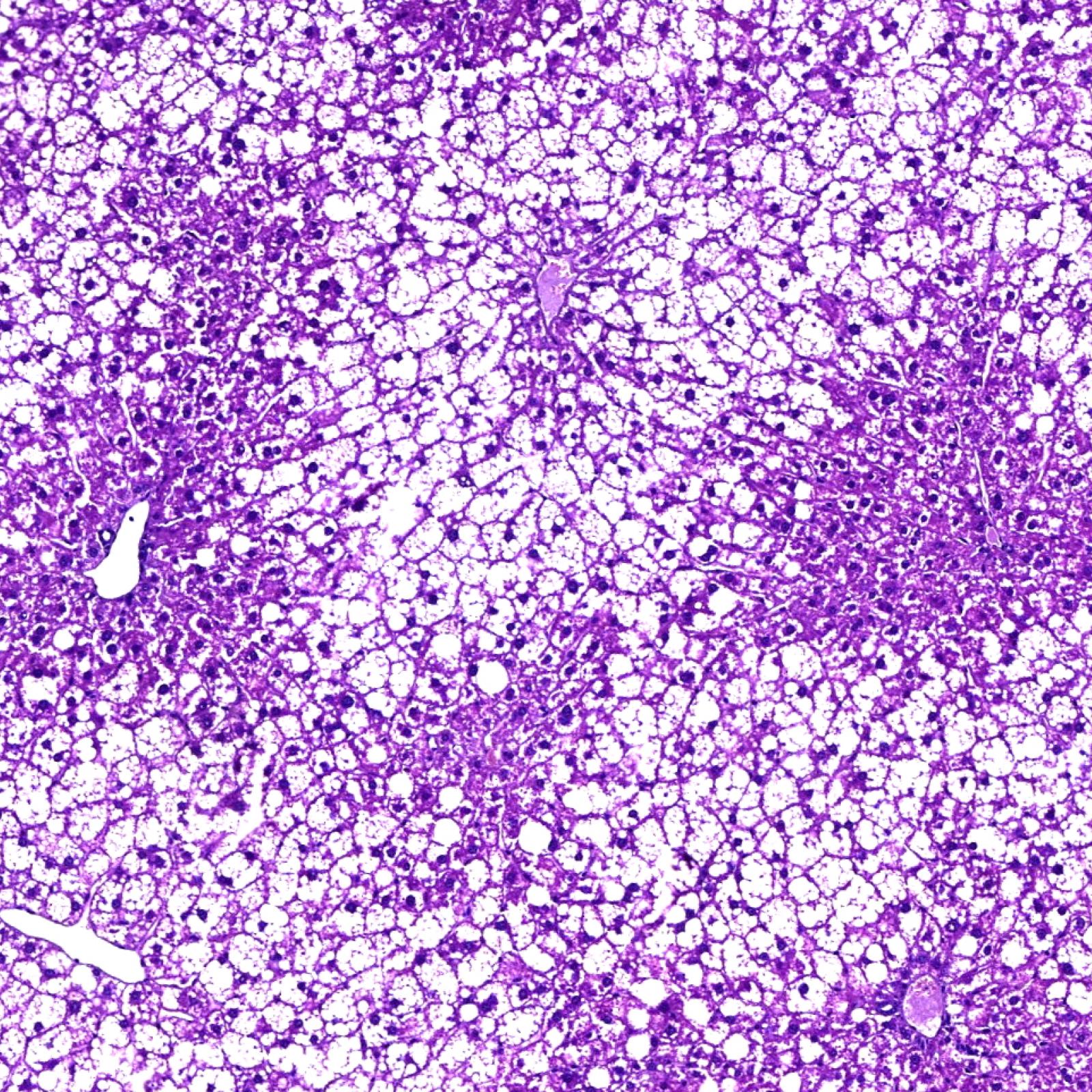
P. Hegde, E. Weiss, V. Paradis, A. Lehuen, S. Lotersztajn
European patent “USE OF AN AGENT CAPABLE OF INHIBITING THE ACTIVATION OF MAIT CELLS FOR THE TREATMENT OF OBESITY AND OBESITY-RELATED DISORDERS”
EP19 305 332.9 on March 19 2019
PCT/EP2020/057368 on March 18 2020 (WO2020/187955) via le site espacenet de l’OEB.
US2022184083 (A1) on 2022-06-16
A.Toubal, A. Lehuen
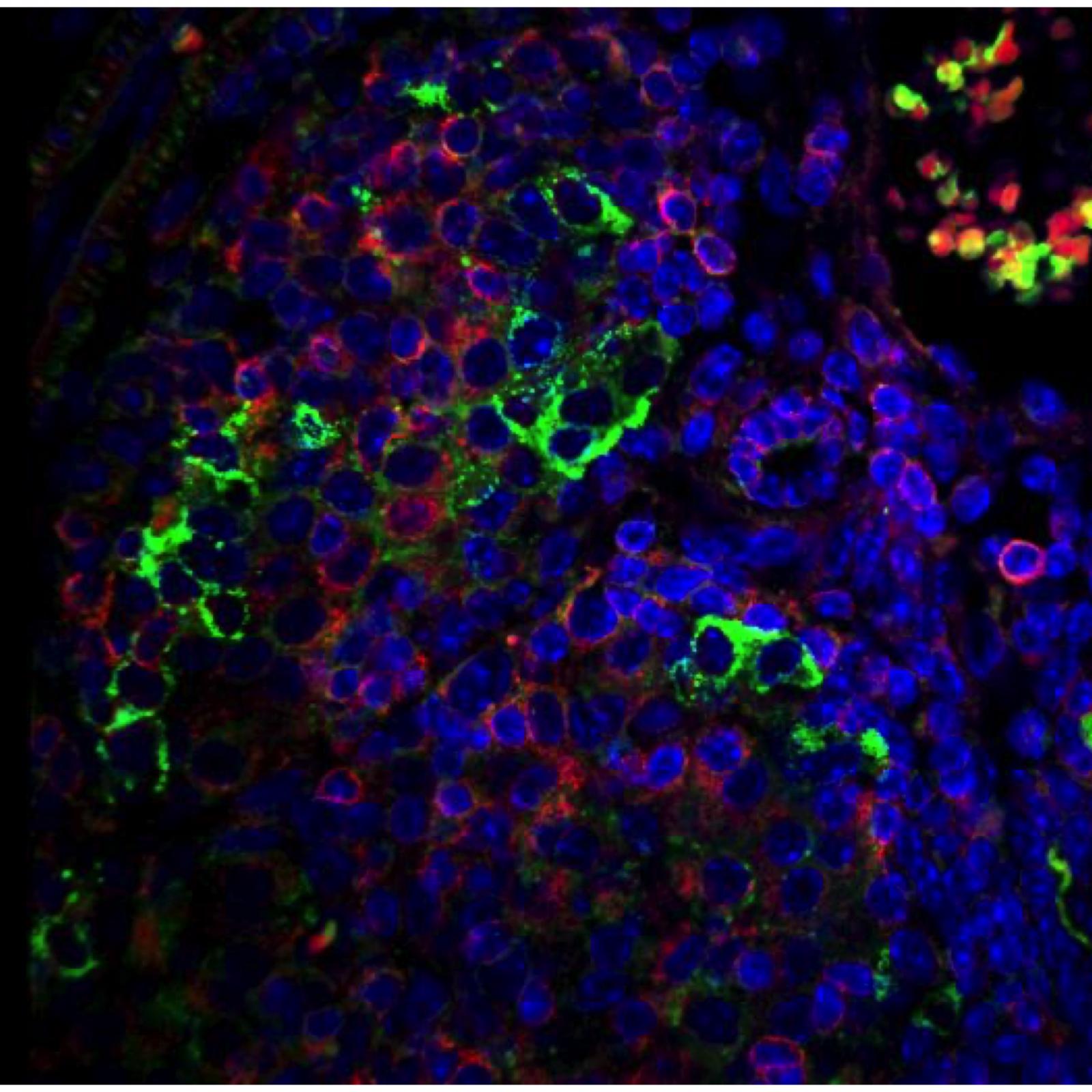
European patent «USE OF MAIT CELLS AS BIOMARKERS AND BIOTARGETS IN COVID-19» filled by Inserm-Transfert
EP20 305 983.7 on September 04 2020
PCT/EP2021/073742 on August 27 2021 (WO2022/043496) via le site espacenet de l’OEB.
H. Flament, M. Rouland, L. Beaudoin, A. Toubal, R.C. Monteiro, A. Lehuen
European patent «USE OF TNF-ALPHA BLOCKING AGENT FOR PREVENTING, SUPPRESSING AND/OR DELAYING THE ONSET OF TYPE 1 DIABETES»
EP21 305 178.2 on February 10 2021
PCT/EP2022/053148 on February 09 2022 (WO2022/171685) accessible online via le site de l’OMPI
M. Rouland, L. Beaudoin, L. Beltrand, A. Lehuen
VALORISATION CLINIQUE
Interactions entre les cellules MAIT, la muqueuse et le microbiome intestinal au cours du développement du diabète de type 1 MAIT, DT1 et microbiome
Périmétre : Régional
Instance, agence publique : INSERM
Niveau d'implication : Coordinateur
Date du début : 01 février 2022
MAIT and SARS-CoV2 with COVID-19 REACTing, COVIDICUS, RADIPEM cohorts
Périmètre : Régional
Instance, agence publique : AP-HP, INSERM, Tours hospital
Niveau d'implication : Coordinateur
Date du début : 05 février 2020
Intitulé : RHU QUID NASH
Périmètre : Régional
Instance, agence publique : AP-HP et INSERM
Niveau d'implication : Partenaire
Date du début : 27 janvier 2018
FINANCEMENTS MAJEURS
2021 - 2024 INSERM Programme Microbiote
2021 - 2025 ANR HepadiMAIT
2020 - 2025 ANR Labex Inflamex
2019 - 2024 Label "Equipe FRM"
2019 - 2024 Award of the Fondation Francophone pour la Recherche sur le Diabète
2019 - 2020 Award of the Juvenile Diabetes Research Foundation
2018 - 2021 ANR Diab1MAIT
2017 - Network Research Hospital-University in Health (RHU QUID NASH)